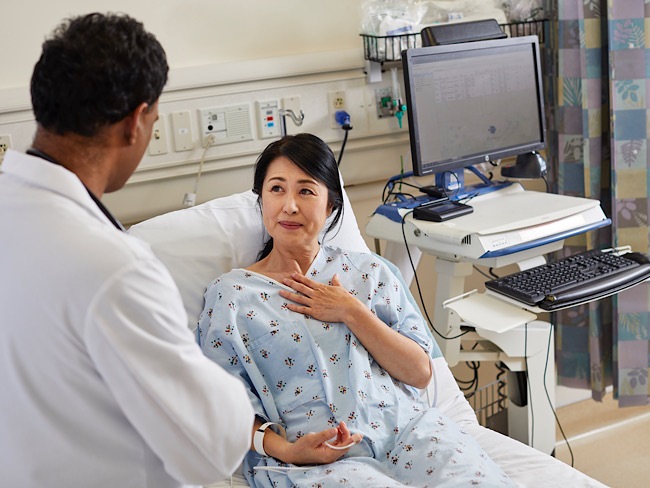
Cervical cancer screening: Exploring the at-home HPV test
Kaiser Permanente is at the forefront of cervical cancer research. Find out how home tests for HPV — a leading cause of cervical cancer — help increase early detection.
Soon, patients may have the option to take easy, convenient tests for HPV in the privacy of their own homes.
Scientific breakthroughs in cancer screening have saved many lives over the past decades. But sometimes the biggest barrier to effective cancer screening isn’t scientific knowledge. It’s what people have to do to get screened.
Take cervical cancer. There are several ways to help prevent cervical cancer. One way is to identify and remove precancers caused by high-risk strains of the human papillomavirus, or HPV.
Deaths from cervical cancer in the United States have declined dramatically over the last 50 years. This is mainly due to screening for HPV in people with cervixes.
But recently screening rates have been declining.
Barriers to cervical cancer screening
One in 4 women don’t receive regular HPV screenings. And half of all diagnosed cervical cancers occur in people who are not screened.
When asked why they miss cervical cancer screenings, people point to many barriers.
Many lack the time and transportation to get to screening appointments. Others say screenings are uncomfortable or embarrassing. And pelvic exams can be triggering for people who have experienced trauma.
Researchers at the Kaiser Permanente Washington Health Research Institute set out to see if more people would get screened if they had access to more convenient options.
What we’re learning could change the landscape of cervical cancer screening for patients everywhere.
Testing for HPV at home
Kaiser Permanente has already boosted colorectal cancer screening rates among our members. Mailing at-home tests to people when they are due for screenings was key to that success.
Using that program as a model, our team conducted a study to see if at-home tests for HPV could increase screening rates for cervical cancer.
The research study included 16,590 people from 30 to 65 years old. We sent participants home kits with everything they needed to collect a sample swab.
They returned their samples to our labs for testing to see if they had the HPV strains most likely to cause cervical cancer. They received their results and follow-up care just like patients who had been screened in one of our clinics.
Our research showed that samples collected at home detect HPV just as well as samples collected by a clinician.
Learning how to improve the experience of at-home HPV testing
Our team also tracked response rates when they mailed HPV testing kits to people who were overdue for cervical cancer screening.
Mailing HPV kits increased screening by more than 50%.
A follow-up survey showed that people who returned the kits had very positive feelings about the experience.
Some people who didn’t return the kits said they felt unsure about how to use them correctly. Others said they didn’t want to insert the swab or felt embarrassed to use the kit.
Our team is working on addressing these challenges in an ongoing study of 33,000 people.
The study explores different outreach approaches based on participants’ prior screening behavior. It also explores whether information included with the test kits can motivate people to get screened.
The future of at-home HPV tests
Although several countries use at-home HPV tests, the U.S. Food and Drug Administration has not yet approved the kits. But research at Kaiser Permanente and other institutions is helping to pave the way for their approval in the United States.
At-home HPV testing kits would give people a more convenient choice for early detection of cervical cancer.
For some people, that choice could save their lives.
Learn more about cancer care at Kaiser Permanente.
-
Social Share
- Share Cervical Cancer Screening: Exploring the At-Home HPV Test on Pinterest
- Share Cervical Cancer Screening: Exploring the At-Home HPV Test on LinkedIn
- Share Cervical Cancer Screening: Exploring the At-Home HPV Test on Twitter
- Share Cervical Cancer Screening: Exploring the At-Home HPV Test on Facebook
- Print Cervical Cancer Screening: Exploring the At-Home HPV Test
- Email Cervical Cancer Screening: Exploring the At-Home HPV Test
March 27, 2025
Living proof: Colon cancer highly treatable if caught early
There is an alarming rise in colon cancer rates among younger adults. Cynthia …

March 25, 2025
AI in health care: 7 principles of responsible use
These guidelines ensure we use artificial intelligence tools that are safe …
March 7, 2025
Kaiser Permanente in Hawaii cures 1,000th patient with hepatitis C
The milestone highlights its Viral Hepatitis Clinic's crucial role in addressing …
March 7, 2025
High blood pressure during pregnancy is on the rise
The keys to preventing cardiovascular conditions during pregnancy are knowing …

February 27, 2025
‘The heart attack that saved my life’
A sudden heart attack led Mark Twichel to seek care at Kaiser Permanente …

February 26, 2025
Colon cancer: Do you need to be screened?
If you’re age 45 or older, getting checked regularly for colorectal cancer …

February 26, 2025
Spring into a better night’s sleep
A Kaiser Permanente sleep expert shares tips for decreasing the impact …

February 20, 2025
Kaiser Permanente joins Food Is Medicine Colorado coalition
As an inaugural member, Kaiser Permanente will help lead health care’s …

February 18, 2025
I just need a chance
Billy Cardosi spent his life putting his family and job first. Then a heart-rela …

February 14, 2025
A fulfilling life on the other side of ovarian cancer
As a wife and a mother, Autumn Gray was determined to beat cancer to be …

February 12, 2025
Back on track after a rare cancer diagnosis
After facing sarcoma at age 18, drag-racing champion Cooper Chun needed …

February 4, 2025
What is therapy, anyway?
At Kaiser Permanente, our care is personalized for each patient's mental …

January 24, 2025
Is one drink a day OK? Here’s what to consider
NPR

January 15, 2025
After surgery, home was where his heart was
Virtual cardiac rehabilitation offers Mike Erskine a convenient, safe way …

January 13, 2025
How to prevent cervical cancer
Cervical cancer is highly preventable. HPV vaccination and regular screenings …

December 26, 2024
How telehealth can make life easier for people with cancer
Virtual care connects cancer patients like Rob Tufel to a wide range of …

November 26, 2024
Personalize your care with a family health history
Talk with your family members about their medical conditions. What you …

November 26, 2024
How to reduce your risk of stroke
A Kaiser Permanente doctor and researcher shares the simple changes you …

November 18, 2024
The power of early detection and proactive men’s health
A father's determination to stay healthy for his 3 children led him to …

November 13, 2024
Self-care for caregivers matters: Here’s why
A sharp increase in rates of adult caregiving is taking a mental and physical …

November 12, 2024
Surviving lung cancer as a nonsmoker
As a lifelong nonsmoker, Mariann Stephens was shocked to learn she had …

October 29, 2024
That’s not tennis elbow
A Kaiser Permanente physician thought he pinched a nerve during a tennis …

October 15, 2024
107-year-old member credits Kaiser Permanente for longevity
Bettye Garrett’s personal doctor says her healthy perspective on life’s …
October 8, 2024
It started with a bad feeling
In August 2023, John Lynn collapsed at work. For the next year, he fought …
October 4, 2024
Teacher learns about herself and how to live with anxiety
A lifelong educator seeks behavioral health care to manage repetitive worries, …

October 1, 2024
Screening for breast cancer: Mammogram guidelines
A Kaiser Permanente radiologist answers commonly asked questions.

October 1, 2024
From depression to connection: Older adult finds her way
After a long period of loneliness and isolation, Roberta Maguire gets the …

September 20, 2024
Ovarian cancer journey confirmed nursing student's calling
Miriam Gutierrez was diagnosed with late-stage ovarian cancer at age 31. …

September 19, 2024
First look at new Lakewood facilities
New medical offices will enhance the health care experience for members …
September 18, 2024
More than 100 ‘Top Docs’ recognized in Washington state
Kaiser Permanente celebrates doctors and nurse practitioners recognized …

September 18, 2024
Cancer rates are rising in younger age groups
‘Connect’ with research to help understand more about the increase of certain …

September 17, 2024
A Latina’s voice in mental health is impactful with her clients
Connecting through a shared heritage gives one therapist a unique perspective …

September 17, 2024
Playing on after 2 decades of cancer care
With the support of his health care team and nurse navigator, musician …

September 17, 2024
Groundbreaking at new medical offices in Pueblo
The new Pueblo North Medical Offices will replace the existing facility …

September 6, 2024
Navigating a world turned upside down after heart failure
After a life-threatening series of heart attacks at age 57, Bunnell Fockler …

August 29, 2024
After Stage 4 ovarian cancer, she’s still going strong
Donna Budway received prompt surgery followed by chemotherapy. She credits …

August 29, 2024
Neurosurgery and a deep, enduring doctor-patient relationship
A basketball coach’s remarkable story of undergoing brain tumor surgery …

August 28, 2024
Making breastfeeding work: A second-time mom’s journey
Support from a dedicated care team helps Tiffany Anonye breastfeed her …

August 27, 2024
From sore throats to ice cream floats
A mother and daughter share a special bond after undergoing tonsillectomies …

August 26, 2024
Katie's ride: Motorcycle rally a surprise for patient
Kaiser Permanente in San Diego helped plan a special send-off for Katie …
August 15, 2024
Back home one day after heart surgery
Ed Dalmasso needed an aortic valve replacement. His care team provided …

August 6, 2024
For a father with prostate cancer, knowledge is power
Harold Newman had advanced prostate cancer. Genetic testing helped expand …

August 1, 2024
Kaiser Permanente tops Colorado ‘Top Doctors’ list
5280 Magazine named 332 Kaiser Permanente physicians to its annual Top …

July 10, 2024
We help members lower their risk of heart attacks and strokes
A Southern California program, powered by our connected care model, is …

July 10, 2024
Grant to help make school lunches healthier for kids
Chef Ann Foundation will use $275,000 grant for Colorado program to convert …

July 2, 2024
Reducing cultural barriers to food security
To reduce barriers, Food Bank of the Rockies’ Culturally Responsive Food …

June 28, 2024
Operation Splash makes a splash for safe summer fun
Kaiser Permanente is making waves this summer, ensuring that communities …

June 28, 2024
Health Action Summit highlights mental health opportunities
The Kaiser Permanente Colorado Health Action Summit gathered nonprofits, …

June 25, 2024
Prompt postpartum care saves a baby’s life
When a newborn was diagnosed with meningitis, a life-threatening infection …
June 17, 2024
That’s a lot of babies!
Delivering multiple babies is difficult, but Kaiser Permanente care teams …

June 17, 2024
A culture of caring eases a cancer journey
Exceptional, personalized radiation oncology care helped Maura Craig treat …

June 13, 2024
Conquered 2 cancers while climbing mountains
Chris Hogan faced kidney cancer and prostate cancer at the same time. He …

June 3, 2024
A call to ‘Connect’ for cancer prevention research
Participate in a study to help uncover the causes of cancer and how to …

May 31, 2024
Stage 4 lung cancer: A story of hope
A young father is enjoying “bonus time” with his kids thanks to new targeted …

May 21, 2024
Surviving stage 4 lung cancer with immunotherapy treatment
Patients like Carol Pitman are living longer thanks to advances in lung …

May 7, 2024
Making cancer care more convenient in Southern California
Kaiser Permanente has opened a new Radiation Oncology Center at the Bellflower …

May 1, 2024
Tacoma ALS clinic recognized for high-quality care
Patients with amyotrophic lateral sclerosis, also called Lou Gehrig’s disease, …
April 23, 2024
We’re rising up to help prevent falls
Kaiser Permanente is committed to finding ways to help reduce falls in …

April 17, 2024
5 common health conditions men don’t like to talk about
Some of the most common conditions affecting men carry a social stigma …

April 10, 2024
For a new mom, talking about her worries helped her heal
One in 5 people experience depression, anxiety, or other mental health …

April 9, 2024
Denver Fire Department annual blood work screenings triple
It’s easy to put off recommended health screenings, and sometimes even …

April 8, 2024
Reducing inequity with fruits and vegetables
Black Americans experience worse health outcomes compared to other populations. …

April 1, 2024
Lynch syndrome: Managing the risk of hereditary colon cancer
Lynch syndrome is a gene mutation that increases colon cancer risk. Learn …

March 29, 2024
Faster recovery: From cardiac scare to exploring Italy
Virtual cardiac rehab helped Mike Kelly heal at home after a life-threatening …

March 20, 2024
Life after cancer: Surviving and thriving
A healthy life after cancer is possible. Learn how Kaiser Permanente helps …

March 19, 2024
Fostering responsible AI in health care
With the right policies and partnerships, artificial intelligence can lead …

March 14, 2024
Healthy kidneys support overall good health
Kaiser Permanente excels in preventing, detecting, and treating kidney …

March 14, 2024
Midwife offers personal care for mom facing complications
For Sam Beeson, having a midwife at her side during her pregnancy helped …
March 12, 2024
In-home recovery restores harmony to life
Colorado musician back with the band thanks to new virtual cardiac rehab …

March 6, 2024
Joining a national effort to test new ways to find cancer
As part of the Cancer Screening Research Network, our researchers will …

March 6, 2024
Colon cancer screening: She’s glad she didn’t wait
A timely preventive test reveals Rebecca Kucera has cancer. Swift treatment …

March 5, 2024
Researchers look for ways to find pancreatic cancer early
Early detection of the disease, before it becomes advanced, will increase …

February 22, 2024
The journey of a lifetime
Care teams at Kaiser Permanente Fontana Medical Center helped Phillip Crawford …

February 21, 2024
From planning his funeral to celebrating his wedding
Gabriel Abarca had no hope for his future. Then the team at Kaiser Permanente …

February 21, 2024
Recovering at home after a double mastectomy
Innovative surgical recovery program helps breast cancer patients safely …

February 1, 2024
Take a break from cannabis while expecting, study suggests
Research shows that marijuana use during pregnancy increases health risks …

January 31, 2024
Prioritizing policies for health and well-being in Colorado
CityHealth’s 2023 Annual Policy Assessment awards cities for their policies …

January 24, 2024
A full-circle journey for one cancer survivor
Grateful for compassionate and successful Hodgkin lymphoma treatment at …

January 17, 2024
How diabetes can affect your heart
People with diabetes are more likely to have heart disease.

January 10, 2024
‘You don’t know unless you ask them’
Kaiser Permanente’s Patient Advisory Councils help us create exceptional …
December 20, 2023
Research transforms care for people with multiple sclerosis
Our researchers are leading the way to more effective, affordable, and …

December 19, 2023
Determined to drop the weight and stop the cycle of diabetes
Following a COVID-19-related hospital stay, Robert DeLeon took charge of …

December 15, 2023
Family-centered care, through pregnancy and beyond
Members experiencing a low risk pregnancy have the option of having their …

December 13, 2023
Nurse navigators guide patients from diagnosis to treatment
An unexpected cancer diagnosis left Jennifer Martin unsure of the next …

December 12, 2023
Hundreds attend first Food Is Medicine Summit
Attendees look at ways to get healthy food to people who don’t have enough …

December 6, 2023
Leading research with gratitude
Learn how you can participate in a study to uncover what causes cancer …

December 6, 2023
Video prenatal visits are a boon for a busy working mom
A new care option offers a mix of in-person and virtual visits, supported …

December 6, 2023
Leaders named among health care’s most influential
Greg A. Adams; Maria Ansari, MD, FACC; and Ramin Davidoff, MD, have been …

December 1, 2023
Surviving — and thriving — after cancer
From diagnosis to recovery, David Parsons, MD, shares how screening, treatment, …

October 25, 2023
Breast cancer during pregnancy: Caring for mom and baby
A team of specialists treats an expecting mother’s cancer while keeping …

October 24, 2023
Childhood anxiety: What parents need to know
A child and adolescent psychiatrist shares tips on supporting your child …

October 23, 2023
A renewed sense of purpose after surviving breast cancer
Joy Short, a Kaiser Permanente member and employee, turned her breast cancer …

October 23, 2023
The future of health care is digital
Nari Gopala, Kaiser Permanente’s chief digital officer, answers 3 questions …

October 11, 2023
Early breast cancer detection improves quality of life
For 75-year-old Peggy Dickston, a surprise diagnosis was caught early thanks …

October 4, 2023
An easier way to manage multiple prescriptions
If you have an ongoing health condition, you know it can be tricky to keep …

October 3, 2023
Nursing excellence recognized at Fontana Medical Center
The prestigious Magnet® designation affirms the compassion, dedication, …

September 27, 2023
10 school districts receive next round of RISE grants
The Thriving Schools program helps educators and students in Colorado integrate …

September 19, 2023
Is telehealth right for you?
Members give video visits high marks — and with a few simple tips, you …

September 13, 2023
Transforming the medical record
Kaiser Permanente’s adoption of disruptive technology in the 1970s sparked …

August 29, 2023
Preventing overdoses starts with education
Risk factors are not always associated with addiction or substance abuse. …